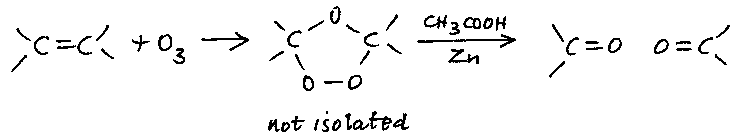 .
.Problem 4
An alcohol has the molecular formula C4H7OH. Oxidation of this alcohol gives a ketone. Ozonolysis of this alcohol gives methanal as one of the products.
Giving your reasoning in full, deduce the structural formula for this alcohol and predict whether it will be optically active.
Ozonolysis is a technique for finding the position of C=C double bonds in a molecule. The compound is reacted with ozone-enriched oxygen in a suitable inert solvent, and the product is hydrolysed with ethanoic acid in the presence of zinc. This produces a mixture of carbonyl compounds, which may be separated and identified, for example by means of their 2,4DNP derivatives or by instrumental (IR, NMR) methods:
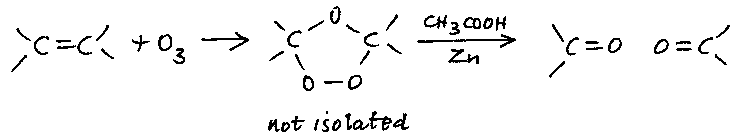 .
.
Answer 4 Organic problems contents Home page